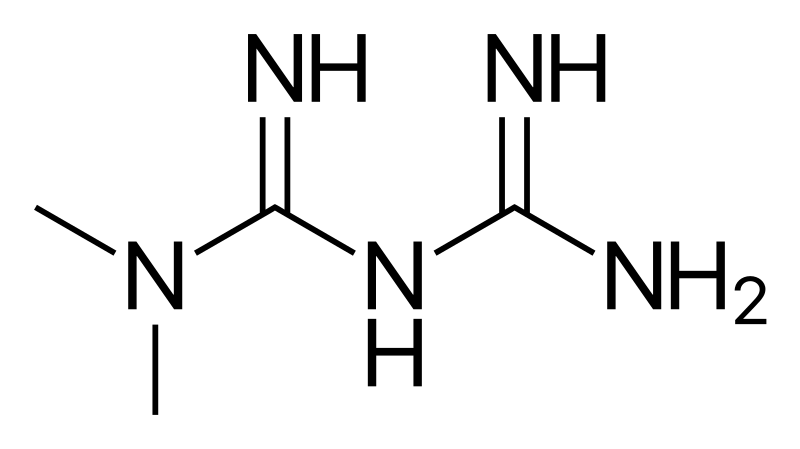
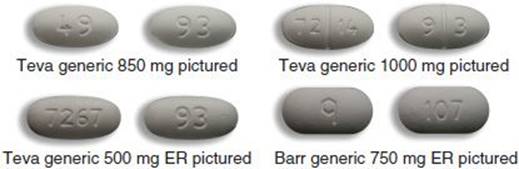
Teva Pharmaceuticals Initiates Voluntary Nationwide Recall of Metformin ER Tablets 500 mg and 750 mg Due to Detection of NDMA

Teva Pharmaceuticals USA, Inc. Initiates Voluntary Nationwide Recall of Metformin Hydrochloride Extended-Release Tablets USP 500 mg and 750 mg Due to Detection of N-Nitrosodimethylamine (NDMA) | FDA